ThyroVet
Product Type: PRESCRIPTION ANIMAL DRUG LABEL
Autor Name: MWI/VetOne
Code Source: 13985-549
Route of Administration: ORAL
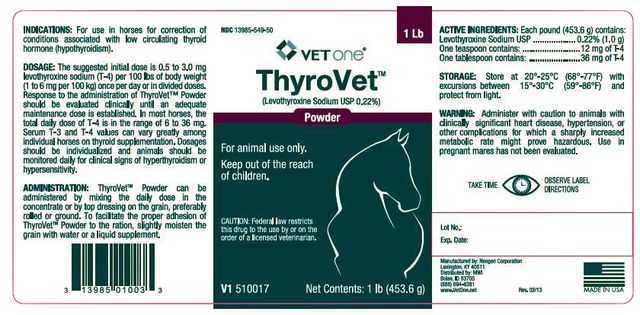
??‹ThyroVet ™
(Levothyroxine Sodium USP 0.22%)
??‹Powder
For animl use only.
Keep out of the reach of children.
CAUTION: Federal law restricts this drug to the use by or on the order of a licensed veterinarian.
Levothyroxine Sodium USP.....0.22% (1.0 g)
One teaspoon contains.........12 mg of T-4
One tablespoon contains.......36 mg of T-4
TAKE TIME
OBSERVE LABEL DIRECTIONS
Lot No.:
Exp. Date:
Manufactured by:
Neogen Corporation
Lexington, KY 40511
Distributed by: MWI
Boise, ID 83705
(888) 694-8381
www.VetOne.net
Rev. 02/13
MADE IN USA
1 Lb
??‹VETone®
??‹ThyroVet ™
(Levothyroxine Sodium USP 0.22%)
??‹Powder
For animl use only.
Keep out of the reach of children.
CAUTION: Federal law restricts this drug to the use by or on the order of a licensed veterinarian.
V1510017
Net Contents: 1 lb (453.6 g)
VETone ThryoVet Powder 1 lb
10 Lb
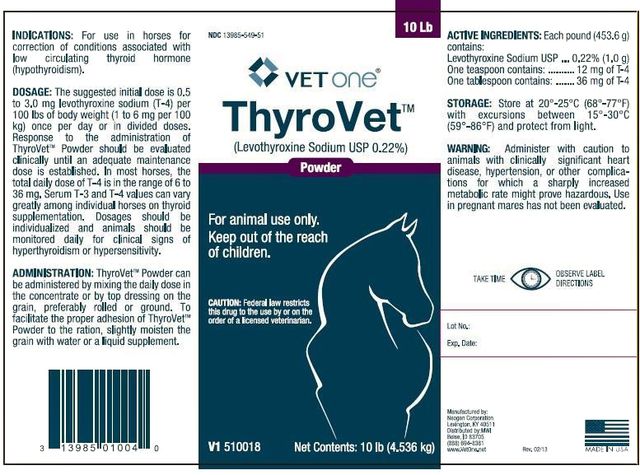
??‹VETone®
??‹ThyroVet ™
(Levothyroxine Sodium USP 0.22%)
Powder
For animal use only.
Keep out of the reach of children.
??‹CAUTION: ??‹Federal law restricts this drug to the use by or on the order of a licensed veterinarian.
??‹V1 ??‹510018
Net Contents: 10 lb (4.536 kg)
VETone ThyroVet Powder 10lb
Autor Name: MWI/VetOne
Code Source: 13985-549
Route of Administration: ORAL
ThyroVet
Generic: Levothyroxine SodiumIngredients:
- LEVOTHYROXINE SODIUM LEVOTHYROXINE : Active ingredient - reference substance is basis of strength - 1 g in 453.6 g
Package Description:
- 453.6 g in 1 BOTTLE, PLASTIC
- 4536 g in 1 PAIL
??‹VETone®
??‹ThyroVet ™
(Levothyroxine Sodium USP 0.22%)
??‹Powder
For animl use only.
Keep out of the reach of children.
CAUTION: Federal law restricts this drug to the use by or on the order of a licensed veterinarian.
For use in horses for correction of conditions associated with low circulating thyroid hormone (hypothyroidism).
The suggested initial dose is 0.5 to 3.0 mg levothyroxine sodium (T-4) per 100 lbs of body weight (1 to 6 mg per 100 kg) once per day or in divided doses. Response to the administration of ThyroVet ™ Powder should be evaluated clinically until an adequate maintenance dose is established. In most horses, the total daily dose of T-4 is in the range of 6 to 36 mg. Serum T-3 and T-4 values can vary greatly among individual horses on thyroid supplementation. Dosages should be individualized and animals should be monitored daily for clinical signs of hyperthyroidism or hypersensitivity.
ThyroVet ™ Powder can be administered by mixing the daily dose in the concentrate or by top dressing on the grain, preferably rolled or ground. To facilitate the proper adhesion of ThyroVet ™ Powder to the ration slightly moisten the grain with water or a liquid supplement.
Each pound (453.6 g) contains:
Levothyroxine Sodium USP.....0.22% (1.0 g)
One teaspoon contains.........12 mg of T-4
One tablespoon contains.......36 mg of T-4
Store at 20 °-25°C (68°-77°F) with excursions between 15°- 30°C (59°-86°F) and protect from light.
Administer with caution to animals with clinically significant heart disease, hypertension, or other complications for which a sharply increased metabolic rate might prove hazardous. Use in pregnant mares has not been evaluated.
TAKE TIME
OBSERVE LABEL DIRECTIONS
Lot No.:
Exp. Date:
Manufactured by:
Neogen Corporation
Lexington, KY 40511
Distributed by: MWI
Boise, ID 83705
(888) 694-8381
www.VetOne.net
Rev. 02/13
MADE IN USA
??‹NDC ??‹13985-549-50
1 Lb
??‹VETone®
??‹ThyroVet ™
(Levothyroxine Sodium USP 0.22%)
??‹Powder
For animl use only.
Keep out of the reach of children.
CAUTION: Federal law restricts this drug to the use by or on the order of a licensed veterinarian.
V1510017
Net Contents: 1 lb (453.6 g)
VETone ThryoVet Powder 1 lb

??‹NDC ??‹13985-549-51
10 Lb
??‹VETone®
??‹ThyroVet ™
(Levothyroxine Sodium USP 0.22%)
Powder
For animal use only.
Keep out of the reach of children.
??‹CAUTION: ??‹Federal law restricts this drug to the use by or on the order of a licensed veterinarian.
??‹V1 ??‹510018
Net Contents: 10 lb (4.536 kg)
VETone ThyroVet Powder 10lb

